
- China University of Science and Technology makes progress in proton exchange membrane fuel cell electrocatalysts
- Fuel cell can still maintain a discharge power of 81.5% after 30,000 cycles durability test
- Ultra-small platinum-based electrocatalysts achieve long-term cycle stability of fuel cells
Recently, the experimental group of Professor Changzheng Wu from the School of Chemistry and Materials Science of the University of Science and Technology of China and the theoretical calculation group of Professor Heng’an Wu from the Department of Modern Mechanics of the School of Engineering Science have collaborated to synthesize ultra-small platinum-based intermetallic electrocatalysts based on the catalyst.
The assembled proton exchange membrane fuel cell can still maintain a discharge power of 81.5% after 30,000 cycles durability test, realize the high power discharge of the fuel cell and long-term cycle stability, and reveal the mass transfer mechanism of the three-phase interface in the battery. The relevant results were published in the Proc. Natl. Acad. Sci. 2021, DOI: 10.1073/pnas. 2104026118 in the journal Proceedings of the National Academy of Sciences (PNAS) in August.
Compared with other types of batteries, the proton exchange membrane fuel cell has the advantages of high discharge power and no pollution. Among them, the cathode oxygen reduction reaction is the rapid control step of the battery’s overall reaction. Platinum-based intermetallic compounds have natural advantages in stability due to their long-range ordered structure, and are the next generation fuel cell commercial oxygen reduction catalyst system. At present, platinum-based intermetallic compounds still have problems such as large particle size, resulting in reduced platinum utilization and mass activity, which has become one of the key bottlenecks restricting the improvement of fuel cell performance.
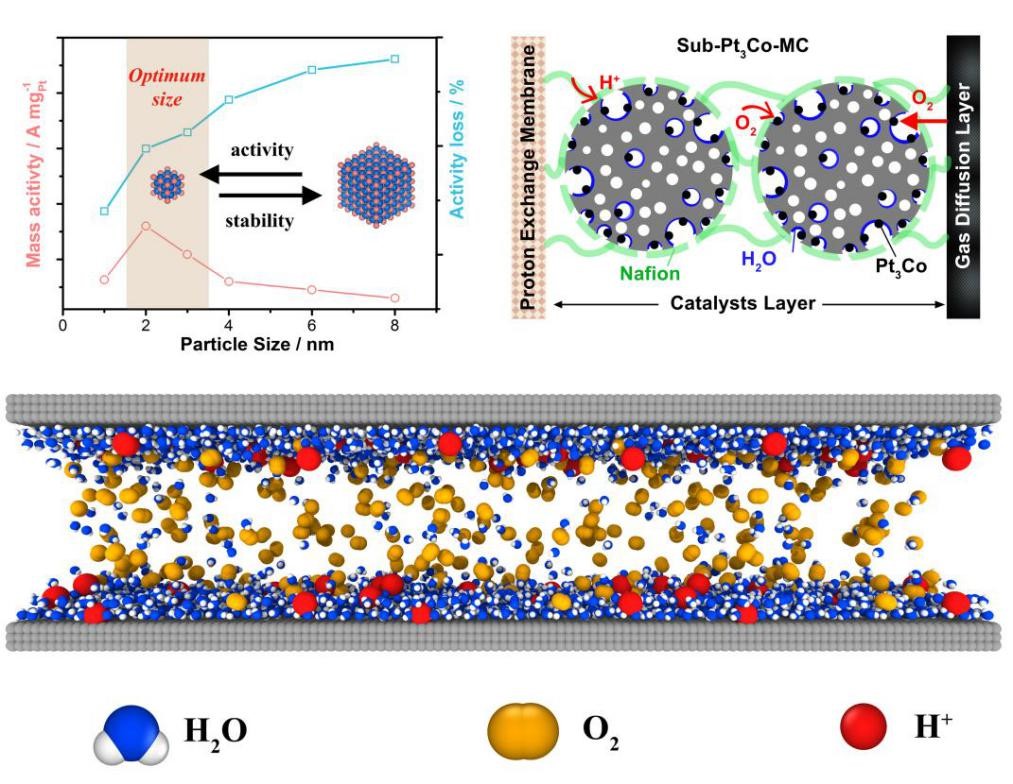
Structural design and optimization of ultra-small platinum-based intermetallic compounds in fuel cell membrane electrodes
In response to this challenge, Professor Changzheng Wu’s team synthesized a series of platinum-based intermetallic compounds Pt3Co, PtCo, and Pt3Ti particles with an ultra-small size of about 2 nm. The power density of the fuel cell assembled from ultra-small Pt3Co intermetallic compound particles is 530 mW/cm-2 higher than that of commercial Pt/C. In the durability test, the mass activity of ultra-small Pt3Co particles can still reach 0.75 A/mg after 30,000 cycles. At the same time, the platinum-based intermetallic compound particles located inside the mesoporous carbon are conducive to the optimization of the three-phase interface under fuel cell operating conditions. Theoretical calculations show that the inside of the mesopores can efficiently complete the transmission of protons and oxygen and achieve dynamic equilibrium, which greatly reduces the mass transfer resistance of the battery, and at the same time prevents the ionomer from poisoning the battery catalyst.
This research optimizes the structure design of the catalyst in the fuel cell membrane electrode based on the ultra-small platinum-based intermetallic compound from the nanometer to the mesoscale system and realizes the high performance expression, providing a new idea for the fuel cell cathode catalyst material.
Cheng Han, postdoctoral fellow in the research group of Professor Wu Changzheng at the University of Science and Technology of China, PhD student Gui Renjie and postdoctoral fellow in the research group of Professor Wu Heng’an Yu Hao are the co-first authors of this paper, and Professor Wu Changzheng is the corresponding author. This research work has been funded by special funds such as the National Key Basic Research and Development Program, the National Natural Science Foundation of China, the China Postdoctoral Science Foundation, and the Anhui Natural Science Foundation.
Link to the paper: https://www.pnas.org/content/118/35/e2104026118
Read the most up to date Fuel Cell and Hydrogen Industry news at FuelCellsWorks




