
As the simplest and most abundant element known to us, the element hydrogen is considered the basic building block of the universe.
On Earth, hydrogen is most commonly found in combination with oxygen in the form of water, covering 71% of the earth’s surface and an essential element for all forms of life on earth.
Versatile and environmentally friendly when produced from renewable energy, hydrogen produces no emissions, and is proving to be a key element in our global efforts to decarbonize electricity, heating, transport and industrial processes. Hydrogen can be safely stored or transported. It can be blended or used to create hydrocarbon fuels. With continued innovation over decades, hydrogen has become an important part of the world’s current alternative power landscape. Cummins uses fuel cell and hydrogen technologies to power a variety of applications, including transit buses, semi-trucks, delivery trucks and passenger trains to name a few. But what exactly are hydrogen fuel cells and how do they work?
Hydrogen fuel cells start in space
Decades ago, hydrogen was used as a fuel in the United States National Aeronautics and Space Administration (NASA) space program, so it is not surprising that, today, hydrogen is used to provide power for systems as large as power supply stations and as small as laptop computers (yes, really!). However, the latest hydrogen technologies also enable the supply of sufficient energy for demanding applications like passenger trains and utility power stations, which demonstrate the many possibilities of using hydrogen as a fuel. Hydrogen fuel cells are seen as a viable alternative technology for automotive, heavy mobility and energy supply industries, complementing battery electric solutions. With water as the only by-product, fuel cells work as an environmentally-friendly power solution.
Hydrogen plus oxygen equals electric energy
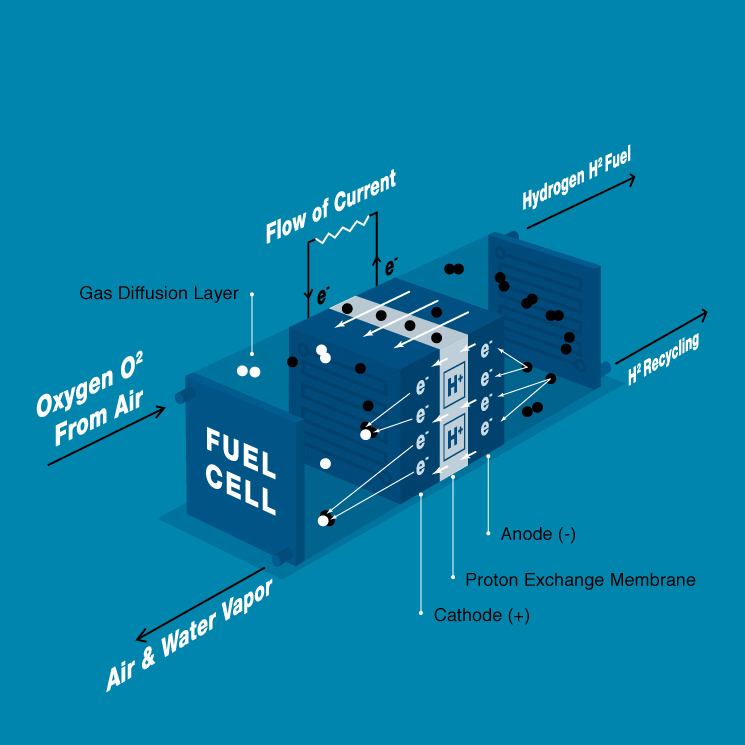 Hydrogen fuel cells are an alternative source of power for a variety of applications. Simply explained, a fuel cell is a device that converts the chemical energy of joining hydrogen molecules with oxygen molecules from the ambient air into electrical energy. The by-product of this reaction is heat and water, which exits the system in the form of water vapor. If you need a refresher, read our top five things you should know about hydrogen fuel cells.
Hydrogen fuel cells are an alternative source of power for a variety of applications. Simply explained, a fuel cell is a device that converts the chemical energy of joining hydrogen molecules with oxygen molecules from the ambient air into electrical energy. The by-product of this reaction is heat and water, which exits the system in the form of water vapor. If you need a refresher, read our top five things you should know about hydrogen fuel cells.
Looking at the process and the architecture of fuel cells in more detail, each single cell consists of four parts: the anode, the cathode, the electrolyte and the gas diffusion layers. Similar to how batteries work, the anode is the negative and a cathode is the positive electrode. In the fuel cell’s case, the anode is fed with pressurized hydrogen and the cathode with air which contains oxygen. The anode and cathode are separated by a special electrolyte membrane in the core of the cell. A catalyst on both the anode and cathode facilitates the splitting of the molecules and enables the reaction of oxygen and hydrogen. As only the hydrogen protons are capable of passing through the membrane, and the electrons are forced to flow from the anode through the load externally, joining up again on the cathode side. This specific type of fuel cell is called a PEM (Proton Exchange Membrane) fuel cell.
The gas diffusion layers facilitate the transport and even spread of the reactants over the entire active area surface. When the hydrogen hits the catalyst, it splits into protons and electrons. Whereas the hydrogen protons can pass the membrane, the electrons are forced to travel through an external circuit before combining with the hydrogen protons and O2 on the other side to form water. The gas diffusion layer helps to remove the water which is formed on the cathode side from the system.
Hydrogen fuel cells powering our applications
In Fuel Cell Electric Vehicle (FCEV) powertrains, the electricity generated by the flow of electrons in the fuel cell can be used to drive an electric motor, generate mechanical power, drive accessories and charge high-voltage battery packs. By stacking multiple fuel cells together, the electric voltage can be increased, and by feeding more air and hydrogen to the cells or by increasing the active surface area of the cells, more current and therefore more power for the application is achieved. The stack is embedded in a module, together with balance of plant (BoP) subsystems, comprising fuel, air and water management, coolant control hardware and software. The fuel cell module can then be integrated into a complete electric power system with suitable cooling units such as radiators, along with power electronics, hydrogen storage tanks and hybrid batteries that can be used in a wide range of applications.
With more than 2,000 fuel cell systems in operation worldwide since 2001, Cummins has proven that fuel cells are a viable solution for powering a wide range of applications, many of which have demanding requirements. In addition to diesel, natural gas and battery electric technologies, Cummins is proud to offer our customers the power of choice and ultimately, the best in class technology for their needs.
Learn more about Cummins’ vision and strategy for hydrogen and fuel cell technologies.
Read the most up to date Fuel Cell and Hydrogen Industry news at FuelCellsWorks




