Fuel cells can now be used to generate hydrogen “on-demand” using new technology developed by researchers at Ariel University in Israel.
Hydrogen is produced in a catalytic hydrolysis reaction of sodium borohydride with ruthenium powder as a catalyst. The proposed generator is portable and lightweight; has high energy density; is easy to use, refill, and clean; and is designed for long working periods with the capability for restart after prolonged rests.
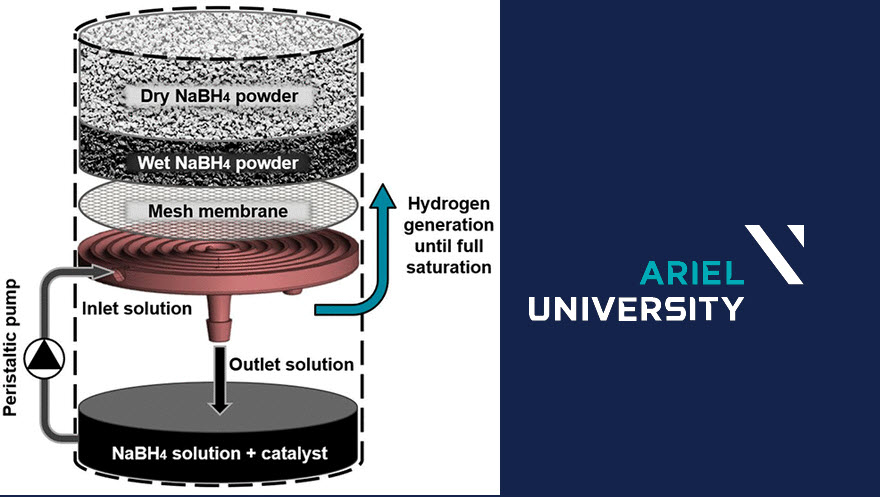
The system consists of two main chambers: the upper chamber with granulated sodium borohydride powder and the lower reaction chamber with a solution of water and catalyst. A peristaltic pump regulates the flow of solution through a spiral channel which is situated in the solids chamber.
The powder is separated from the channel by a mesh which prevents its clogging. The solution becomes saturated while flowing through the channel and drips into the reaction chamber. This system is significantly lighter and requires much less operating energy than other similar generators.
To maintain constant H2 flow rate levels, the solution temperature and H2 pressure in the reaction chamber are constantly controlled. Experiments were carried out to prove system reliability and efficiency, demonstrating multiple stop-and-run breaks at different solution concentrations.
During 6.3 h of the experiment, 110 L of hydrogen was generated with an average flow rate of 290 mL/min and 98% conversion efficiency. The achieved energy density is 1300 Wh/kg for the fuel, 540 Wh/kg for the generator, and 377 Wh/kg with the attached 30 W fuel cell. This hydrogen generation has a great potential for a variety of military, vehicle, and portable device applications.
Lev Zakhvatkin, Michael Zolotih, Yonatan Maurice, Alex Schechter, and Idit Avrahami (2021) “Hydrogen Production on Demand by a Pump Controlled Hydrolysis of Granulated Sodium Borohydride” Energy & Fuels doi: 10.1021/acs.energyfuels.1c00367
Read the most up to date Fuel Cell and Hydrogen Industry news at FuelCellsWorks




